Clarifoam EF
Generic name:sulfur,sulfacetamide sodium
Dosage form: aerosol, foam
Drug class:Topical antibiotics
Medically reviewed by Drugs.com. Last updated on May 23, 2022.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
On This Page
Clarifoam EF Description
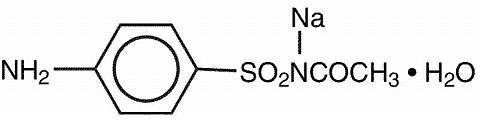
Sodium sulfacetamide is a sulfonamide with antibacterial activity while sulfur acts as a keratolytic agent. Chemically, sodium sulfacetamide is N-[(4-aminophenyl) sulfonyl]- acetamide, monosodium salt, monohydrate.
The structural formula is:
Each gram of Clarifoam® EF (sodium sulfacetamide 10% and sulfur 5%) Emollient Foam contains 100 mg of sodium sulfacetamide and 50 mg of sulfur in an aqueous based emollient foam vehicle containing cetyl alcohol NF, emulsifying wax NF, lactic acid USP, methylparaben NF, propylene glycol USP, propylparaben NF, steareth-10, water USP. Also contains: Propellant HFA-134A (1,1,1,2-tetrafluoroethane).
Clarifoam EF - Clinical Pharmacology
Sodium sulfacetamide exhibits antibacterial activity. The most widely accepted mechanism of action of sulfonamides is the Woods-Fildes theory which is based on the fact that sulfonamides act as competitive antagonists to para-aminobenzoic acid (PABA), an essential component for bacterial growth. While absorption through intact skin has not been determined, sodium sulfacetamide is readily absorbed from the gastrointestinal tract when taken orally and excreted in the urine, largely unchanged. The biological half-life has variously been reported as 7 to 12.8 hours.
It is estimated that 1% of topically applied sulfur is absorbed. Although the exact mode of keratolytic activity of sulfur is unknown, it is reported to result from the interaction of sulfur with the cysteine content of keratinocytes. In combination with sulfacetamide, sulfur has been reported to inhibit the growth of Propionibacterium acnes thereby reducing the associated inflammation.