Dextrose Injection
Generic name: dextrose monohydrate
Dosage form: injection, solution
Drug class:Glucose elevating agents
Medically reviewed by Drugs.com. Last updated on Feb 1, 2022.
On This Page
Rx only
DESCRIPTION:
5% Dextrose Injection, USP solution is sterile and nonpyrogenic. It is a parenteral solution containing dextrose in water for injection intended for intravenous administration.
Each 100 mL of 5% Dextrose Injection, USP, contains dextrose monohydrate, 5 g in water for injection. The caloric value is 170 kcal/L. The osmolarity is 252 mOsmol/L (calc.), which is slightly hypotonic.
The solution pH is 4.3 (3.2 to 6.5).
This solution contains no bacteriostat, antimicrobial agent or added buffer and is intended only as a single-dose injection. When smaller doses are required the unused portion should be discarded.
5% Dextrose Injection, USP is a parenteral fluid and nutrient replenisher.
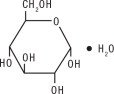
Dextrose, USP is chemically designated D-glucose monohydrate (C 6H 12O 6 • H 2O), a hexose sugar freely soluble in water. It has the following structural formula:
Water for Injection, USP is chemically designated H 2O.
The flexible container is fabricated from a specially formulated non-plasticized film containing polypropylene and thermoplastic elastomers (freeflex® bag). The freeflex® + bag has a needle-free injection port and can accept standard luer lock syringes to add medication. The amount of water that can permeate from the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the flexible container can leach out certain of the container's chemical components in very small amounts within the expiration period. The suitability of the container material has been confirmed by tests in animals according to USP biological tests for plastic containers.
CLINICAL PHARMACOLOGY:
When administered intravenously, these solutions provi...